Suggestions
Use up and down arrows to review and enter to select.Please wait while we process your payment
If you don't see it, please check your spam folder. Sometimes it can end up there.
If you don't see it, please check your spam folder. Sometimes it can end up there.
Please wait while we process your payment

By signing up you agree to our terms and privacy policy.
Don’t have an account? Subscribe now
Create Your Account
Sign up for your FREE 7-day trial
Already have an account? Log in
Your Email
Choose Your Plan
Individual
Group Discount
Save over 50% with a SparkNotes PLUS Annual Plan!
 payment page
payment page
Purchasing SparkNotes PLUS for a group?
Get Annual Plans at a discount when you buy 2 or more!
Price
$24.99 $18.74 /subscription + tax
Subtotal $37.48 + tax
Save 25% on 2-49 accounts
Save 30% on 50-99 accounts
Want 100 or more? Contact us for a customized plan.
 payment page
payment page
Your Plan
Payment Details
Payment Summary
SparkNotes Plus
You'll be billed after your free trial ends.
7-Day Free Trial
Not Applicable
Renews May 2, 2024 April 25, 2024
Discounts (applied to next billing)
DUE NOW
US $0.00
SNPLUSROCKS20 | 20% Discount
This is not a valid promo code.
Discount Code (one code per order)
SparkNotes PLUS Annual Plan - Group Discount
Qty: 00
SparkNotes Plus subscription is $4.99/month or $24.99/year as selected above. The free trial period is the first 7 days of your subscription. TO CANCEL YOUR SUBSCRIPTION AND AVOID BEING CHARGED, YOU MUST CANCEL BEFORE THE END OF THE FREE TRIAL PERIOD. You may cancel your subscription on your Subscription and Billing page or contact Customer Support at custserv@bn.com. Your subscription will continue automatically once the free trial period is over. Free trial is available to new customers only.
Choose Your Plan
For the next 7 days, you'll have access to awesome PLUS stuff like AP English test prep, No Fear Shakespeare translations and audio, a note-taking tool, personalized dashboard, & much more!
You’ve successfully purchased a group discount. Your group members can use the joining link below to redeem their group membership. You'll also receive an email with the link.
Members will be prompted to log in or create an account to redeem their group membership.
Thanks for creating a SparkNotes account! Continue to start your free trial.
We're sorry, we could not create your account. SparkNotes PLUS is not available in your country. See what countries we’re in.
There was an error creating your account. Please check your payment details and try again.
Please wait while we process your payment

Your PLUS subscription has expired
Please wait while we process your payment
Please wait while we process your payment

Problem :
3.0 moles of gas at 300 K and 1.0 atm have a volume of 15 smoots. What is
R in 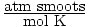 ?
?
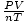 = R. Plug in the values
of P, V, n, and T to solve for R. R = 0.017
= R. Plug in the values
of P, V, n, and T to solve for R. R = 0.017 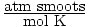 .
.
Problem : A sample of gas at 1.0 atm and 298 K has a volume of 8.7 L. How much gas does the sample contain?
Rearrange PV = nRT to solve for n.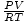 = n. Let's try
converting everything to SI units so that we can used the gas constant value
of 8.314
= n. Let's try
converting everything to SI units so that we can used the gas constant value
of 8.314 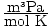 . 1.0 atm converts to 1.0×105 Pa. There are 10-3 m3 in a liter, so 8.7 L converts to
8.7×10-3 m3. Plugging in these values, we find that n = 0.35
mol.
. 1.0 atm converts to 1.0×105 Pa. There are 10-3 m3 in a liter, so 8.7 L converts to
8.7×10-3 m3. Plugging in these values, we find that n = 0.35
mol.
Problem : You are the head engineer at a chemical plant. Sam Yagan, the summer intern, has just broken the cooling system to the main helium tank. Sam tells you not to be worried; the tank can withstand up to 40.0 atm of pressure before exploding. Before Sam's accident, the tank was subjected to 10.0 atm. The tank was originally at 250 K, but it's a hot summer day, and you fear its temperature may rise to 350 K. Should you believe Sam?
The key to this problem is to realize which values are constant. V, n, and R do not change. P and T do. Rearrange PV = nRT so that the variables and constants are on opposite sides of the "=" sign: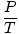 = = 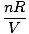 = C = C |
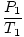 = = 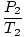 |
Problem : A race of purely gas-based aliens inhabits Jupiter (P = 808 kPa, T = 600 K). On Jupiter, each alien occupies a volume of one cubic meter. The aliens decide to visit Earth. They board their climate controlled gascraft, make the long journey to Earth, and land in a cornfield in southern Idaho (P = 101 kPa, T = 300 K ). When they disembark, how big will they be? Assume that the aliens' innards do not diffuse or burst in the Earth's atmosphere.
P, T, and V are the variables in this problem. n is constant. Place the constants and variables on opposite sides of the "=" sign: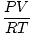 = n = n |
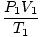 = = 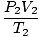 →V2 = →V2 = 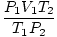 |
Please wait while we process your payment

