Problem :
If the percent yield of the following equation is 55%,
how many moles of H2SO4 are produced when 44.8 L
of SO3 are combined with excess water?
| SO3(g) + H2O(l)→H2SO4(aq) |
|
The equation is balanced, so convert to moles.
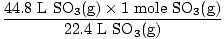 = 2 moles SO3(g) = 2 moles SO3(g) |
|
Now calculate the theoretical yield. How many moles of
H2SO4(aq) should we get?
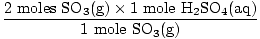 = 2 moles H2SO4(aq) = 2 moles H2SO4(aq) |
|
Now solve algebraically for the actual yield.
| 55% = 0.55 = Actual Yield/(2 moles H2SO4(aq)) | | |
|
| 2 moles H2SO4(aq)×0.55 = 1.1 moles H2SO4(aq) | | |
|