-
Average Value
The Average Value of a Property A is the value expected if the measurement were taken on every system in the ensemble and averaged, and is written as < A >.
-
Closed System
A system that maintains a constant number of particles, constant energy, constant volume, and is free from any change in influences external to the system, such as an oscillating magnetic field.
-
Conventional
Conventional is a term applied to variables in common uses in the natural sciences and chemistry, but which in turn are defined in relation to their fundamental counterparts. We use the term to apply to T and S, the conventional temperature and entropy. Compare to fundamental.
-
Ensemble
A collection of isolated systems, all of which are identical to each other.
-
Entropy
Entropy, represented by σ, is a variable that describes the randomness of a system and is defined by σ(N, U)âÉálog g(N, U).
-
Fundamental
Fundamental is a term applied to a variable to indicate that it is naturally defined. We use the term to apply to σ and τ, the entropy and temperature. Compare to conventional.
-
Fundamental Assumption
The Fundamental Assumption states that any closed system has an equal probability to be in any of its possible quantum states.
-
Laws of Thermodynamics
The 4 Laws of Thermodynamics are postulates upon which the study of thermodynamics historically rested. They exist in numerous formulations that at times appear unrelated to each other, but can be better understood from a statistical approach.
-
Multiplicity Function
The Multiplicity Function g(N, U) yields the number of states with the same values of N and U.
-
State
The minimal collection of information about a system that is maximally informative.
-
Temperature
Temperature, represented by τ, is a variable that describes the thermal agitation of a system and is defined by
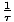 =
= 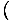
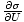
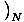 .
.
Terms
Formulae

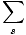 A(s)P(s)
A(s)P(s)


 payment page
payment page



