Please wait while we process your payment
If you don't see it, please check your spam folder. Sometimes it can end up there.
If you don't see it, please check your spam folder. Sometimes it can end up there.
Please wait while we process your payment
Get instant, ad-free access to our grade-boosting study tools with a 7-day free trial!
Learn more



This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.
Create Account
Select Plan
Payment Info
Start 7-Day Free Trial!

Annual
2-49 accounts
$22.49/year + tax
50-99 accounts
$20.99/year + tax
Select Quantity
Price per seat
$29.99 $--.--
Subtotal
$-.--
Want 100 or more? Request a customized plan
You could save over 50%
by choosing an Annual Plan!

SAVE OVER 50%
compared to the monthly price!
| Focused-studying | ||
| PLUS Study Tools | ||
| AP® Test Prep PLUS | ||
| My PLUS Activity | ||
$22.49/month + tax
Save 25%
on 2-49 accounts
$20.99/month + tax
Save 30%
on 50-99 accounts
| Focused-studying | ||
| PLUS Study Tools | ||
| AP® Test Prep PLUS | ||
| My PLUS Activity | ||
No Fear provides access to Shakespeare for students who normally couldn’t (or wouldn’t) read his plays. It’s also a very useful tool when trying to explain Shakespeare’s wordplay!
Erika M.
I tutor high school students in a variety of subjects. Having access to the literature translations helps me to stay informed about the various assignments. Your summaries and translations are invaluable.
Kathy B.
Teaching Shakespeare to today's generation can be challenging. No Fear helps a ton with understanding the crux of the text.
Kay H.
No Fear provides access to Shakespeare for students who normally couldn’t (or wouldn’t) read his plays. It’s also a very useful tool when trying to explain Shakespeare’s wordplay!
Erika M.
I tutor high school students in a variety of subjects. Having access to the literature translations helps me to stay informed about the various assignments. Your summaries and translations are invaluable.
Kathy B.
Teaching Shakespeare to today's generation can be challenging. No Fear helps a ton with understanding the crux of the text.
Kay H.
Create Account
Select Plan
Payment Info
Start 7-Day Free Trial!
You will only be charged after the completion of the 7-day free trial.
If you cancel your account before the free trial is over, you will not be charged.
You will only be charged after the completion of the 7-day free trial. If you cancel your account before the free trial is over, you will not be charged.
Order Summary
Annual
7-day Free Trial
SparkNotes PLUS
$29.99 / year
Annual
Quantity
51
PLUS Group Discount
$29.99 $29.99 / seat
Tax
$0.00
SPARK25
-$1.25
25% Off
Total billed on Nov 7, 2024 after 7-day free trail
$29.99
Total billed
$0.00
Due Today
$0.00
Promo code
This is not a valid promo code
Card Details
By placing your order you agree to our terms of service and privacy policy.
By saving your payment information you allow SparkNotes to charge you for future payments in accordance with their terms.
Powered by stripe
Legal
Google pay.......



Please wait while we process your payment

Sorry, you must enter a valid email address
By entering an email, you agree to our privacy policy.
Please wait while we process your payment

Sorry, you must enter a valid email address
By entering an email, you agree to our privacy policy.
Please wait while we process your payment

Your PLUS subscription has expired
Please wait while we process your payment
Please wait while we process your payment

Nucleotides and Nucleic Acids
Both DNA and RNA are known as nucleic acids. They have been given this name for the simple reason that they are made up of structures called nucleotides. Those nucleotides, themselves comprising a number of components, bond together to form the double-helix first discovered by the scientists James Watson and Francis Crick in 1956. This discovery won the two scientists the Nobel Prize. For now, when we discuss nucleic acids you should assume we are discussing DNA rather than RNA, unless otherwise specified.
A nucleotide consists of three things:
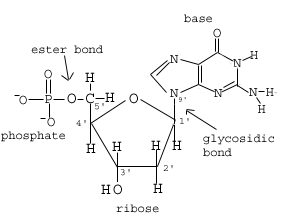
Nucleotides join together through phosphodiester linkages between the 5' and 3' carbon atoms to form nucleic acids. The 3' -OH of the sugar group forms a bond with one of the negatively charged oxygens of the phosphate group attached to the 5' carbon of another sugar. When many of these nucleotide subunits combine, the result is the large single-stranded polynucleotide or nucleic acid, DNA ()

If you look closely, you can see that the two sides of the nucleic acid strand shown above are different, resulting in polarity. At one end of the large molecule, the carbon group is unbound and at the other end, the -OH is unbound. These different ends are called the 5'- and 3'-ends, respectively.
shows a single strand of DNA. However, as stated earlier,
DNA exists as a double-helix, meaning two strands of DNA bind together.
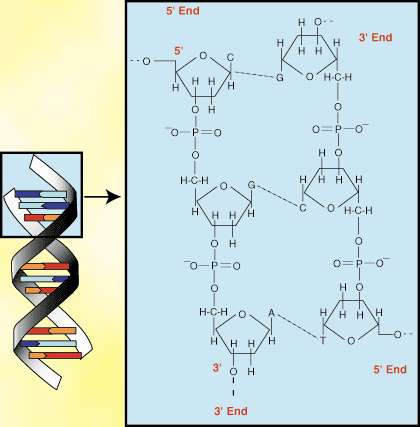
An important thing to remember about the structure of the DNA helix is that as a result of anti-parallel pairing, the nitrogen base groups face the inside of the helix while the sugar and phosphate groups face outward. The sugar and phosphate groups in the helix therefore make up the phosphate backbone of DNA. The backbone is highly negatively charged as a result of the phosphate groups.
Please wait while we process your payment





