Please wait while we process your payment
If you don't see it, please check your spam folder. Sometimes it can end up there.
If you don't see it, please check your spam folder. Sometimes it can end up there.
Please wait while we process your payment

By signing up you agree to our terms and privacy policy.
Don’t have an account? Subscribe now
Create Your Account
Sign up for your FREE 7-day trial
By signing up you agree to our terms and privacy policy.
Already have an account? Log in
Your Email
Choose Your Plan
Individual
Group Discount
Save over 50% with a SparkNotes PLUS Annual Plan!
 payment page
payment page
Purchasing SparkNotes PLUS for a group?
Get Annual Plans at a discount when you buy 2 or more!
Price
$24.99 $18.74 /subscription + tax
Subtotal $37.48 + tax
Save 25% on 2-49 accounts
Save 30% on 50-99 accounts
Want 100 or more? Contact us for a customized plan.
 payment page
payment page
Your Plan
Payment Details
Payment Summary
SparkNotes Plus
You'll be billed after your free trial ends.
7-Day Free Trial
Not Applicable
Renews May 7, 2025 April 30, 2025
Discounts (applied to next billing)
DUE NOW
US $0.00
SNPLUSROCKS20 | 20% Discount
This is not a valid promo code.
Discount Code (one code per order)
SparkNotes PLUS Annual Plan - Group Discount
Qty: 00
SparkNotes Plus subscription is $4.99/month or $24.99/year as selected above. The free trial period is the first 7 days of your subscription. TO CANCEL YOUR SUBSCRIPTION AND AVOID BEING CHARGED, YOU MUST CANCEL BEFORE THE END OF THE FREE TRIAL PERIOD. You may cancel your subscription on your Subscription and Billing page or contact Customer Support at custserv@bn.com. Your subscription will continue automatically once the free trial period is over. Free trial is available to new customers only.
Choose Your Plan
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.
For the next 7 days, you'll have access to awesome PLUS stuff like AP English test prep, No Fear Shakespeare translations and audio, a note-taking tool, personalized dashboard, & much more!
You’ve successfully purchased a group discount. Your group members can use the joining link below to redeem their group membership. You'll also receive an email with the link.
Members will be prompted to log in or create an account to redeem their group membership.
Thanks for creating a SparkNotes account! Continue to start your free trial.
We're sorry, we could not create your account. SparkNotes PLUS is not available in your country. See what countries we’re in.
There was an error creating your account. Please check your payment details and try again.
Please wait while we process your payment

Your PLUS subscription has expired
Please wait while we process your payment
Please wait while we process your payment

Racemic Mixtures and Enantiomeric Excess
A solution in which both enantiomers of a compound are present in equal amounts is called a racemic mixture, or racemate. Racemic mixtures can be symbolized by a (d/l)- or ()- prefix in front of the substance's name. Since enantiomers have equal and opposite specific rotations, a racemic mixture exhibits no optical activity. Therefore it is impossible to tell a racemic mixture apart from an achiral substance using polarimetry alone. Note that the terms chiral and optically active should not be confused. It would be incorrect to say that a racemic mixture is achiral. Chirality is a property of individual molecules. Optical activity is a property of solutions. A racemic mixture consists of chiral molecules, but it has no net optical activity.
The process by which a racemic mixture is formed from chiral materials is called racemization. One way to do this is to mix equal amounts of enantiomeric substances. From this point of view, it may be puzzling that racemic mixtures are important. After all, what are the chances of obtaining any mixture in which two enantiomers are present in exactly equal amounts? It turns out that racemic mixtures actually occur with considerable frequency. Racemic mixtures are often formed when achiral substances are converted into chiral ones. This is due to the fact that chirality can only be distinguished in a chiral environment. An achiral substance in an achiral environment has no preference to form one enantiomer over another.
The separation of enantiomers poses a special problem for chemists. Enantiomers have the same boiling points, melting points, solubilities, etc., so many of the techniques used to separate other compounds don't work on racemic mixtures. The answer to this problem is to separatee nantiomers in a chiral environment, where they interact differently.
One technique is to use a chiral resolving agent. This technique relies on the fact that while enantiomers have identical physical properties, diastereomers generally have different properties. For example, suppose we wanted to separate the enantiomers of 2-hydroxylpropionic acid. We add as the resolving agent an enantiomerically pure amount of (R)-2-phenyl-ethylamine. The two enantiomers interact with (R)-2-phenyl-ethylamine to form two distinct salt species that are diastereomers of each other. The diastereomers can then be crystallized separately.
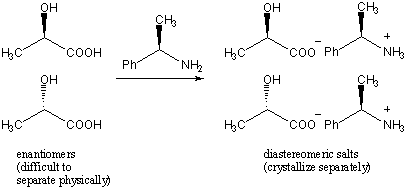
Another technique is to use chiral chromatography. In this process, the racemate is run through a column that is filled with a chiral substance. The enantiomers will interact differently with the substance and will then elute (or filter through the substance) at different rates. These techniques are also applied to mixtures of enantiomers beside racemic mixtures, for example to purify a species from small amounts of its enantiomer.
How important is it for chemists to isolate pure enantiomers? In some applications, the chirality of a molecule is unimportant. In many cases, however, the chirality of a molecule is crucial to its function. This is especially true in biological systems, where a molecule might have a function vastly different from that of its enantiomer. Biological systems are chiral environments. Here are a few examples:
Please wait while we process your payment

