Up until now, all our reactions have taken place under ideal
conditions. However, ideal conditions don't exist in real life.
Reactants may be impure, reactions may not go to completion, or given
reactions may have to compete with several smaller side reactions. In
fact, in the laboratory, if you get 60% of the expected amount of
product, that is considered very good.
Now, let's talk terminology. The calculated or expected amount of
product is called the theoretical yield. The amount of product
actually produced is called the actual yield. When you divide
actual yield by theoretical yield you get a decimal percentage known as
the percent yield of a reaction.
Percent Yield = 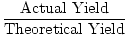 |
|
Once again it's time for an example problem:
Problem: What is the percent yield of the following reaction if
60 grams of CaCO3 is heated to give 15 grams of CaO?
Solution: Ideally, how many grams of CaO should be produced?
First verify the equation is balanced; it is. Now convert to moles,
based on the amount of
CaCO3 present.
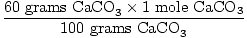 × ×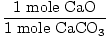 = 0.6 mole CaO = 0.6 mole CaO |
|
How many grams is this?
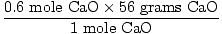 = 33.6 grams CaO = 33.6 grams CaO |
|
So, ideally, 33.6 grams of CaO should have been produced in this
reaction. This is the theoretical yield. However, the problem tells
us that only 15 grams were produced. 15 grams is the actual yield. It
is now a simple matter to find percent yield.
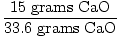 = 0.446 = 44.6% = 0.446 = 44.6% |
|