Gibbs Factor
We derived in Section 1 the Boltzmann factor by
allowing the temperature to change. If we generalize and allow the
number of particles to change as well, we can obtain a ratio of
probabilities:
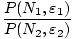
=
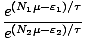
The structure of the above is very similar to what we encountered
before. Ignoring the new term returns the familiar form from before.
Any term of the form e(Nμ- )/τ is called a Gibbs
factor.
)/τ is called a Gibbs
factor.
Gibbs Sum
So far we only have a mechanism for relative probabilities. To obtain
absolute probabilities, we need to have a sum that adds up all of the
Gibbs factors. This sum is called the Gibbs sum and is given by:
Note that in taking the sum we begin with N = 0, which has its own
corresponding energies 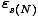 , and sum over all the states
with N = 0. Then we move to N = 1, and so on.
, and sum over all the states
with N = 0. Then we move to N = 1, and so on.
We can calculate the absolute probability of occupation of a state now
using the Gibbs factor and the Gibbs sum just as we did with the
Boltzmann factor and sum earlier.