Suggestions
Use up and down arrows to review and enter to select.Please wait while we process your payment
If you don't see it, please check your spam folder. Sometimes it can end up there.
If you don't see it, please check your spam folder. Sometimes it can end up there.
Please wait while we process your payment

By signing up you agree to our terms and privacy policy.
Don’t have an account? Subscribe now
Create Your Account
Sign up for your FREE 7-day trial
Already have an account? Log in
Your Email
Choose Your Plan
Individual
Group Discount
Save over 50% with a SparkNotes PLUS Annual Plan!
 payment page
payment page
Purchasing SparkNotes PLUS for a group?
Get Annual Plans at a discount when you buy 2 or more!
Price
$24.99 $18.74 /subscription + tax
Subtotal $37.48 + tax
Save 25% on 2-49 accounts
Save 30% on 50-99 accounts
Want 100 or more? Contact us for a customized plan.
 payment page
payment page
Your Plan
Payment Details
Payment Summary
SparkNotes Plus
You'll be billed after your free trial ends.
7-Day Free Trial
Not Applicable
Renews May 8, 2024 May 1, 2024
Discounts (applied to next billing)
DUE NOW
US $0.00
SNPLUSROCKS20 | 20% Discount
This is not a valid promo code.
Discount Code (one code per order)
SparkNotes PLUS Annual Plan - Group Discount
Qty: 00
SparkNotes Plus subscription is $4.99/month or $24.99/year as selected above. The free trial period is the first 7 days of your subscription. TO CANCEL YOUR SUBSCRIPTION AND AVOID BEING CHARGED, YOU MUST CANCEL BEFORE THE END OF THE FREE TRIAL PERIOD. You may cancel your subscription on your Subscription and Billing page or contact Customer Support at custserv@bn.com. Your subscription will continue automatically once the free trial period is over. Free trial is available to new customers only.
Choose Your Plan
For the next 7 days, you'll have access to awesome PLUS stuff like AP English test prep, No Fear Shakespeare translations and audio, a note-taking tool, personalized dashboard, & much more!
You’ve successfully purchased a group discount. Your group members can use the joining link below to redeem their group membership. You'll also receive an email with the link.
Members will be prompted to log in or create an account to redeem their group membership.
Thanks for creating a SparkNotes account! Continue to start your free trial.
We're sorry, we could not create your account. SparkNotes PLUS is not available in your country. See what countries we’re in.
There was an error creating your account. Please check your payment details and try again.
Please wait while we process your payment

Your PLUS subscription has expired
Please wait while we process your payment
Please wait while we process your payment

The pH of a weak base solution is calculated in the same manner as that of a weak acid solution, using Kb instead of a Ka.
To calculate the pH of a mixture of acids in aqueous solution, first decide which acid has the lowest pKa. Calculate the pH as if the strongest acid were the only one in solution. We can ignore the contributions to the pH of the weaker acids because they will be minor in comparison to that of the strongest acid in the group. The exact solution to such a problem requires more complex mathematics and will not be covered in this SparkNote.
A salt of a strong acid and a strong base (such as NaCl from HCl and NaOH) produces a neutral solution when dissolved in water. However, when a salt of a weak acid and a strong base (e.g. NaAc from acetic acid and NaOH), a strong acid and a weak base (e.g. NH4Cl from ammonia and HCl), or a weak acid and a weak base (NH4Ac) is dissolved in water, the solution does not have a neutral pH. These phenomena are explained by the reaction of the salts of weak acids and weak bases with water in hydrolysis reactions. As shown in , these hydrolysis reactions produce the parent weak acids and weak bases of the salts:
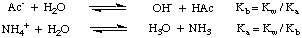
As you can see in the figure above, the salt of a weak acid, such as acetate ion, acts as a base in water, and the salt of a weak base, such as ammonium ion, acts as a weak acid. From our previous discussion on the reactions of acids and bases with water in Disassociations, you should know that the Kb of acetate ion can be calculated from the Ka of acetic acid, and that the Ka of ammonium ion can be calculated from the Kb of ammonia, as shown in .
Another type of hydrolysis reaction comes from the reaction of metal ions with high charges. Such ions act as Lewis acids to water molecules, as shown in . A metal ion can bond to water by accepting an lone pair from the oxygen of a water molecule, and this increases the acidity of water molecule. Like other acids, we can calculate the Ka and calculate the pH of a solution containing such ions.
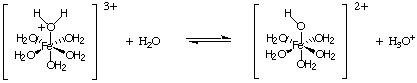
To calculate the pH of a solution containing the salt of a weak acid or a weak base, treat the problem exactly as you did when calculating the pH of weak acid and base solutions above in Calculating pH's, Heading . Mixtures of salts of weak acids and weak bases present a challenging mathematical problem that we will not cover in our treatment of acid-base chemistry.
Please wait while we process your payment

