Please wait while we process your payment
If you don't see it, please check your spam folder. Sometimes it can end up there.
If you don't see it, please check your spam folder. Sometimes it can end up there.
Please wait while we process your payment

By signing up you agree to our terms and privacy policy.
Don’t have an account? Subscribe now
Create Your Account
Sign up for your FREE 7-day trial
By signing up you agree to our terms and privacy policy.
Already have an account? Log in
Your Email
Choose Your Plan
Individual
Group Discount
Save over 50% with a SparkNotes PLUS Annual Plan!
 payment page
payment page
Purchasing SparkNotes PLUS for a group?
Get Annual Plans at a discount when you buy 2 or more!
Price
$24.99 $18.74 /subscription + tax
Subtotal $37.48 + tax
Save 25% on 2-49 accounts
Save 30% on 50-99 accounts
Want 100 or more? Contact us for a customized plan.
 payment page
payment page
Your Plan
Payment Details
Payment Summary
SparkNotes Plus
You'll be billed after your free trial ends.
7-Day Free Trial
Not Applicable
Renews July 6, 2025 June 29, 2025
Discounts (applied to next billing)
DUE NOW
US $0.00
SNPLUSROCKS20 | 20% Discount
This is not a valid promo code.
Discount Code (one code per order)
SparkNotes PLUS Annual Plan - Group Discount
Qty: 00
SparkNotes Plus subscription is $4.99/month or $24.99/year as selected above. The free trial period is the first 7 days of your subscription. TO CANCEL YOUR SUBSCRIPTION AND AVOID BEING CHARGED, YOU MUST CANCEL BEFORE THE END OF THE FREE TRIAL PERIOD. You may cancel your subscription on your Subscription and Billing page or contact Customer Support at custserv@bn.com. Your subscription will continue automatically once the free trial period is over. Free trial is available to new customers only.
Choose Your Plan
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.
For the next 7 days, you'll have access to awesome PLUS stuff like AP English test prep, No Fear Shakespeare translations and audio, a note-taking tool, personalized dashboard, & much more!
You’ve successfully purchased a group discount. Your group members can use the joining link below to redeem their group membership. You'll also receive an email with the link.
Members will be prompted to log in or create an account to redeem their group membership.
Thanks for creating a SparkNotes account! Continue to start your free trial.
We're sorry, we could not create your account. SparkNotes PLUS is not available in your country. See what countries we’re in.
There was an error creating your account. Please check your payment details and try again.
Please wait while we process your payment

Your PLUS subscription has expired
Please wait while we process your payment
Please wait while we process your payment

Atoms and Atomic Orbitals
Magnetic Quantum Number (m): Gives the orientation of the orbital
in space; in other words, the value of m describes whether an orbital
lies along the x-, y-, or z-axis on a three-dimensional graph, with the
nucleus
of the atom at the origin. m can take on any value from -l to l.
For our purposes, it is only important that this quantum number tells us that
for each value of n there may be up to one s-orbital, three p-orbitals, five d-
orbitals, and so on:
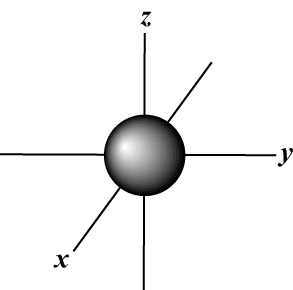
The s orbital (l = 0) has one orbital, since m can only equal 0.
That orbital is spherically symmetrical about the nucleus.
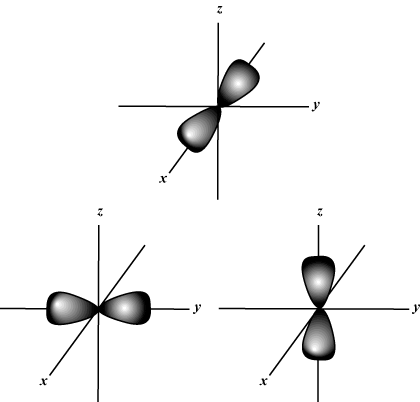
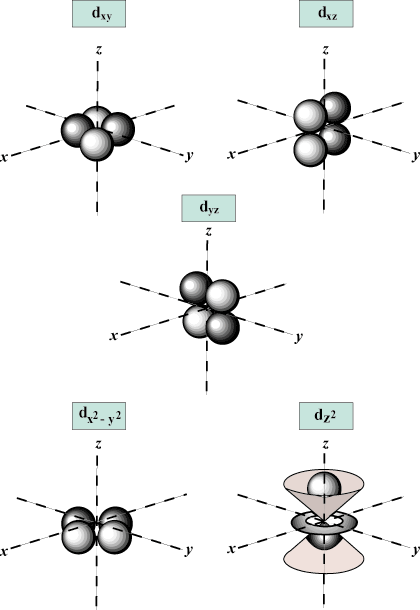
Spin Quantum Number (s): Tells whether a given electron is
spin up (+1/2) or spin down (-1/2). Because the Pauli Exclusion
Principle tells us that no two electrons of an atom can have the same set
of quantum numbers, each orbital is limited to holding two electrons at
most.
It is often convenient to depict orbitals in an orbital energy diagram, as seen below in . Such diagrams show the orbitals and their electron occupancies, as well as any orbital interactions that exist. In this case we have the orbitals of the hydrogen atom with electrons omitted. The first electron shell (n = 1) contains just the 1s orbital. The second shell (n = 2) holds a 2s orbital and three 2p orbitals. The third shell (n = 3) holds one 3s orbital, three 3p orbitals, and five 3d orbitals, and so forth. Note that the relative spacing between orbitals becomes smaller for larger n. In fact, as n gets large the spacing becomes infinitesimally small.

You will see such energy diagrams quite often in your continuing study of organic chemistry. Notice that all orbitals with the same n have the same energy. Orbitals with identical energies are said to be degenerate (not in the moral sense!). Electrons in higher level orbitals have more potential energy and are more reactive, i.e. more likely to undergo chemical reactions.
When an atom only contains a single electron, its orbital energies depend only on the principle quantum numbers: a 2s orbital would be degenerate with a 2p orbital. However, this degeneracy is broken when an atom has more than one electron. This is due to the fact that the attractive nuclear force any electron feels is shielded by the other electrons. s-orbitals tend to be closer to the nucleus than p-orbitals and don't get as much shielding, and hence become lower in energy. This process of breaking degeneracies within a shell is known as splitting. In general s orbitals become lowest in energy, followed by p orbitals, d orbitals, and so forth.

Please wait while we process your payment

