Fermi Gas
A Fermi gas is a gas consisting of fermions. We will
investigate a few of its properties and applications here.
Fermi-Dirac Distribution Function
Consider a system that is a single orbital for a fermion. Remember that
only 0 or 1 fermions may be in the orbital. The Gibbs
sum is easy to calculate then; in fact, we
already solved this problem in a previous problem set:
ZG = 1 + λe- /τ. We can then directly calculate
f, remembering that it is equivalent to < N >.
/τ. We can then directly calculate
f, remembering that it is equivalent to < N >.
f (
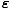
) =
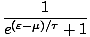
Notice that in the classical limit, f must be much less than 1, so the
exponential must be large compared to one. An expansion yields the
classical distribution function we derived before.
Fermi Level
We are concerned with the case where the gas is dense compared to the
quantum concentration. The gas is called degenerate in this regime,
as opposed to in the classical regime already discussed.
The chemical potential in general is a function of the temperature.
However, we define the chemical potential at a temperature of zero for a
fermi gas to be μ(τ = 0) = 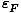 where
where 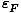 is
called the Fermi energy.
is
called the Fermi energy.
The significance of the Fermi energy is that for τ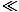
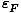 ,
the orbitals of energy
,
the orbitals of energy 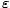 ≤
≤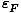 are completely
occupied and the orbitals above the Fermi energy are completely vacant.
are completely
occupied and the orbitals above the Fermi energy are completely vacant.