Energy and the Partition Function
We can relate the partition function to the total energy of the system. Recall that we
can determine the average value of a property
by using the
probability. Let us investigate < 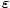 >, the average value of the energy of an
occupied state. It turns out that <
>, the average value of the energy of an
occupied state. It turns out that < 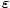 > is precisely what we mean when we
say U. We can calculate it using the above equation for the absolute probability:
> is precisely what we mean when we
say U. We can calculate it using the above equation for the absolute probability:
The final result seems strange but is mathematically tidier and is equivalent to
the messier formulation.
Finally, and significantly, we can relate the Helmholtz free energy, sometimes
just called the free energy, to Z as well. By a simple substitution, F = U - τσ,
we find an important result:
F = - τ log Z