This should make sense. A system wants to minimize its total energy,
and the fermions would pack into the lowest energy states first. If the
temperature is low, there is little thermal excitation to promote any
fermions to orbitals with higher energy.
The Ground State of a Fermi Gas
We can solve for the Fermi energy by setting the total number of
particles below that energy equal to the total number of particles in
the system. We obtain:
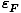
=
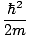
(3
Π2n)
2/3
We use the term "ground state" to refer to the state in which no
fermions are excited to higher energy states beyond the Fermi energy.
We can calculate the energy of the ground state by summing up the
energies of the orbitals below the Fermi energy, to obtain:
Ugs =
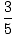 N
N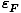
We can go through and calculate all of the other relevant quantities
just as we did for the ideal gas.
The Fermi gas appears throughout any study of physics. Electrons form a
Fermi gas. The electrons in a metal, the "sea of electrons", act as a
Fermi gas. In astrophysics, white dwarf stars are prevented from
collapsing upon themselves by the pressure of the Fermi gas and the
resistance it gives to having its orbitals pushed together.
Bose Gas
A Bose gas is a gas consisting of bosons. We will treat the
topic briefly as above with the Fermi gas.